|
Here E is the total energy of the solid (it has a negative value). The vertical ionization energy is the energy change corresponding to an ionization reaction leading to formation of the ion in a configuration which is the same. In this case, the proportionality constant, k, equals 8. the cohesive properties follow predominantly from the chemical binding in a perfect. The ions are more stable when they are bonded than they were. The equation can also be written using the charge of each ion, expressed in coulombs (C), incorporated in the constant. Then the positive sodium ion and negative chloride ion attract each other and form an ionic bond.  
This value of \( k\) includes the charge of a single electron (1.6022 × 10 −19 C) for each ion. The proportionality constant k is equal to 2.31 × 10 −28 J The general equation for net potential energy EN, for two ions that are separated by the distance r, is as follows: A 8 Furthermore the expression for the equilibrium separation ro (as derived in Problem 2.18) is as follows: In addition, the bonding energy, Eo, is described by the following equation: E0 A 1/ (1 - n) n8 -A n/ (1 - n) For a K+-C. Question: Calculate the bonding energy E0 in terms of the parameters A, B, and n using the following procedure: 1. 2.6 PRIMARY INTERATOMIC BONDS IONIC BONDING Perhaps ionic bonding is the. Interpretation: holding one atom at the origin, a second atom would repel that atom at separation r < roand attract it when ro< r. 3s (b) E lectron energy 2p states for the first three 3.4 n2 2s shells of. 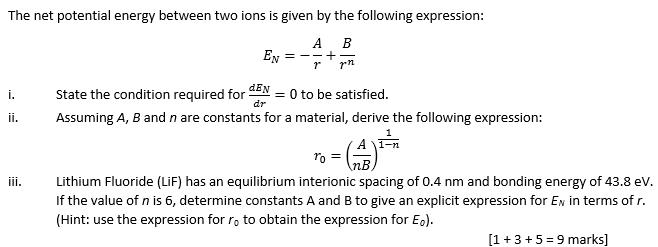
Where each ion’s charge is represented by the symbol \( Q\) and the internuclear distance between the particles is represented by (\(r\)). Bonding energy E0corresponds to the energy at rothe energy that would be required to separate these two atoms to an infinite separation. The internuclear distance in the gas phase is 175 pm.Īnswer: −3180 kJ/mol = −3.\( \newcommand \] Expert Answer 94 (64 ratings) Transcribed image text: 2.18 The net potential energy between two adjacent ions, EN,may be represented hi the sum ofEquations 2.9 and 2.11 that is.EN A/r+B/rnCalculate the bonding energy E0 in terms of the parameters A, B, and n using the following procedure:1. Consequently, in accordance with Equation 4.1.1, much more energy is released when 1 mol of gaseous Li +F − ion pairs is formed (−891 kJ/mol) than when 1 mol of gaseous Na +Cl − ion pairs is formed (−589 kJ/mol).Ĭalculate the amount of energy released when 1 mol of gaseous MgO ion pairs is formed from the separated ions. Ionic bonds In the last section, we looked separately at how sodium can lose an electron to form the cation Na + + and at how chlorine can gain an electron to form the anion Cl. 
Calculate the bonding energy E0 in terms of the parameters A, B, and n using the following procedure: (1) Differentiate EN with respect to r, and then set the. \( \newcommand/mol\right )=-891\ kJ/mol \) īecause Li + and F − are smaller than Na + and Cl − (see Figure 3.2.7 ), the internuclear distance in LiF is shorter than in NaCl. The net potential energy between two adjacent ions, EN, may be represented by EN -A/r + B/rn Where A, B, and n are constants whose values depend on the particular ionic system.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
